| 商品編號: | ECFG21 |
| 商品名稱: | 細胞融合儀 |
| 品牌廠牌: | NepaGene |
| 商品特點: | 細胞融合; 核置換; 四倍體生成; Anti-cancer immunity |
ECFG21 Super Electro Cell Fusion Generator
細胞融合儀
Hybridoma Production, Somatic-Cell Nuclear Transfer and More

全世界第一台自動讀出輸出電壓與電流的細胞融合儀.
細胞融合的效率比傳統 PEG 方法高出非常多.
應用:
• Hybridoma Production
lymphocytes and myeloma cells for monoclonal antibodies
*a large volume chamber is available.
• Somatic-Cell Nuclear Transfer (SCNT)
enucleated oocytes and somatic cells for animal cloning
• Tetraploid embryo complementation
two-cell embryos for ES-cell-derived mice
• Anti-cancer immunity
dendritic cells and tumor cells for cancer vaccines
• Others
* ES/EG cells and thymocytes for nuclear reprogramming of somatic cells
* plant protoplasts for hybrid vegetables
* liposome/droplets
* yeasts/fungi
* cell-culture transfection (electroporation)
| ECFG21 Hybridoma Production for Monoclonal Antibodies |
The fusion efficiency achieved by the ECFG21 for hybridoma production is dramatically higher than PEG. New electrode chambers correspond to a large volume solution (one-mouse lymphocytes).
Electro Cell Fusion Process
| 1) | Alternating Current (AC) |
|
The AC is applied so that the cells are aligned in a chain for touching on each side: "Pearl Chain" Formation |
|
| 2) | Direct Current (DC) Pulses for Fusion |
|
The aligned cells are high-efficiently fusioned by the 2-step DC pulses with voltage decay. |
|
| 3) | Post-Fusion AC |
|
The post-fusion AC is applied so that the fusion process begins to mature and high fusion efficeincy is achieved with low cell damage. |
| ECFG21 vs PEG Small Molecule Antigens or Peptide Antigens |
The efficiency by the ECFG21 is 25-100 times higher than PEG.
These three antigens were all classified as difficult to obtain positive clones. The ECFG21 vastly increased the number of clones in all cases, and in at least one case, resulted in positive clones when none resulted with the traditional PEG method.
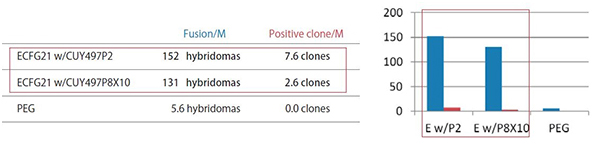
1) Antigen used: 20 kDa immunosuppressive protein with high homology
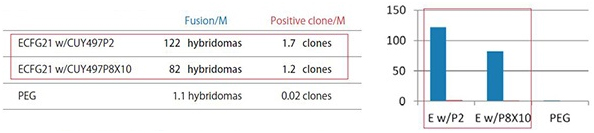
2) Antigen used: Hydrophobic octapeptide
3) Antigen used: Highly hydrophobic 17 amino acid peptide
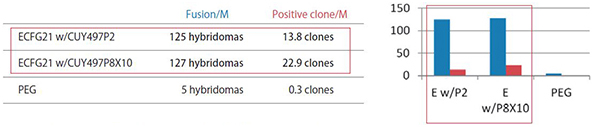
| Fusion/M: | Number of hybridomas generated per 1 x 10^6 lymphocytes |
|
Positive clone/M: |
Number of clones producing monoclonal antibodies against the antigen per 1 x 10^6 lymphocytes |
| ECFG21 w/CUY497P2: |
Electrofusion performed using CUY497P2 electrode (for up to 5 x 10^7 lymphocytes) |
| ECFG21 w/CUY497P8X10: |
Electrofusion performed using CUY497P8X10 electrode (for large volumes: one-mouse lymphocytes) |
| ECFG21 Tetraploid Chimera Production |
| Reprogramming of a melanoma genome by nuclear transplantation |
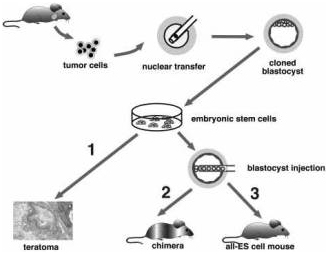
Two-step cloning procedure to produce mice from cancer cells. Different tumor cells were used as donors for nuclear transfer into enucleated oocytes. Resultant blastocysts were explanted in culture to produce ES cell lines. The tumorigenic and differentiation potential of these ES cells was assayed in vitro by inducing teratomas in SCID mice (1), and in vivo by injecting cells into diploid (2) or tetraploid (3) blastocysts to generate chimeras and entirely ES-cell-derived mice, respectively.
Genes Dev. 2004 Aug 1;18(15):1875-85. Hochedlinger K et al., Whitehead Institute for Biomedical Research, and Department of Biology, Massachusetts Institute of Technology
| ECFG21 Liposome/droplet Fusion |
| Timing controllable electrofusion device for aqueous droplet-based microreactors |

High speed camera images of the fusion process This fusion process is almost instantaneous. The two droplets combined into one single ''peanut-shaped'' droplet within about 1ms. It took about another 5ms for the droplet to adopt a spherical shape under the effect of surface tension. Throughout the fusion process, the darker colored blue ink droplet (leftmost) was distinctly separated from the lighter colored water droplet (rightmost).
Lab Chip. 2006 Jun;6(6):757-63. Epub 2006 Mar 31. Tan WH, Takeuchi S., CIRMM/IIS, Institute of Industrial Science, University of Tokyo
| ECFG21 Nuclear Transfer for Animal Cloning |
| Generation of cloned calves and transgenic chimeric embryos from bovine embryonic stem-like cells |

A: Two days after birth B: Four weeks after birth C: Fingerprinting of DNA from cloned calves, recipient cows, and donor ES-like W3 cells. Electrophoretograms show amplified fragments of DNA derived from leukocytes from recipient cows (panels a, c and e) and cloned calves (panels b, d and f) and from donor ES-like W3 cells (panel g). Upper and right-side scales indicate the sizes of DNAs (bp) and the intensities of DNA fragments, respectively. Numbers in boxes indicate the sizes of DNAs (upper) and the intensities of DNA fragments (lower). After insertion of donor ES-like cells into the perivitelline space of oocytes, cells and cytoplasts were fused electrically in fusion medium. (DC: 20V, Pulse length: 50us, Pulse interval: 100ms, 2 Pulses)
Biochem Biophys Res Commun. 2003 Sep 12;309(1):104-13. Saito S et al., Saito Laboratory of Cell Technology, Japan
規格:
* Alternate Current (AC)
| Voltage | 0 - 80 Vrms |
| Frequency | 1 MHz |
| Duration | 0 - 99 sec |
| Post-Fusion Duration | 0 - 99 sec |
| Post-Fusion Decay Mode | On/Off |
| Pause between AC / DC | 5 μsec |
* Direct Current (DC) Pulse
| Pulse Wave | Square Wave |
| Voltage | 1 - 1500 V |
| Pulse Length | 1 - 99 μsec |
| Pulse Interval | 0.1 - 9.9 sec |
| Number of Pulses | 0 - 99 |
| Decay Rate | 0 - 99 % |
| Polarity Switching | On/Off |
* Output Measurements
| Voltage | AC / DC |
| Current | AC / DC |
| Energy (J) | DC |
* Others
| Impedance Measurement | 0.01 - 50 kΩ |
| Operation Mode | Automatic / Manual |
| Memory | 99 programs |
| Dimensions / Weight | 386W x 370D x 121H mm / 8.6kg |
All features and specifications subject to change without notice.
訊詢單內容